 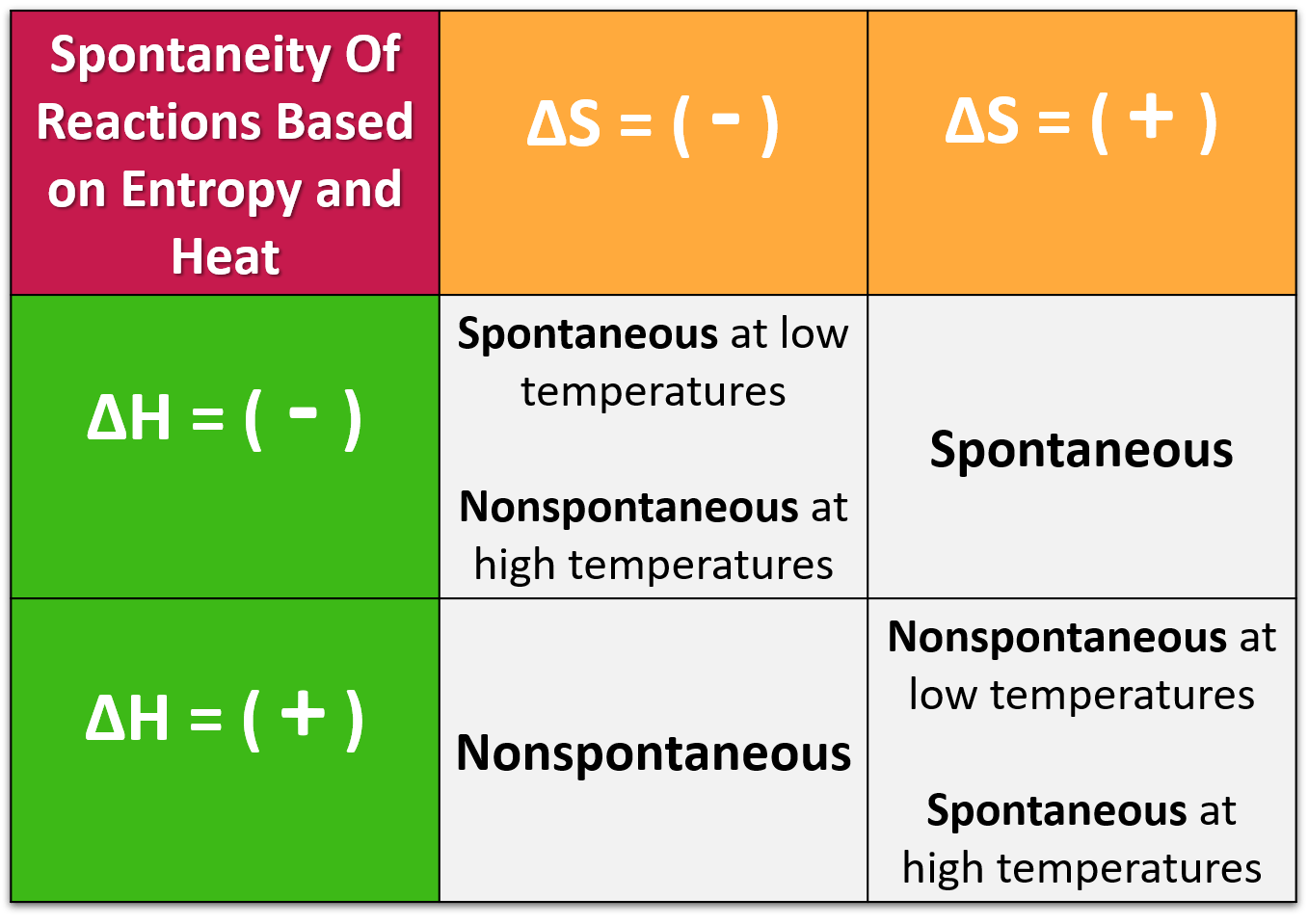
For a given physical process, the entropy of the system and the environment will remain a constant if the process can be reversed. I'm going to stop here before presenting more to see if this initial part of the development is along the lines that you were seeking. The change in entropy (delta S) is equal to the heat transfer (delta Q) divided by the temperature (T). 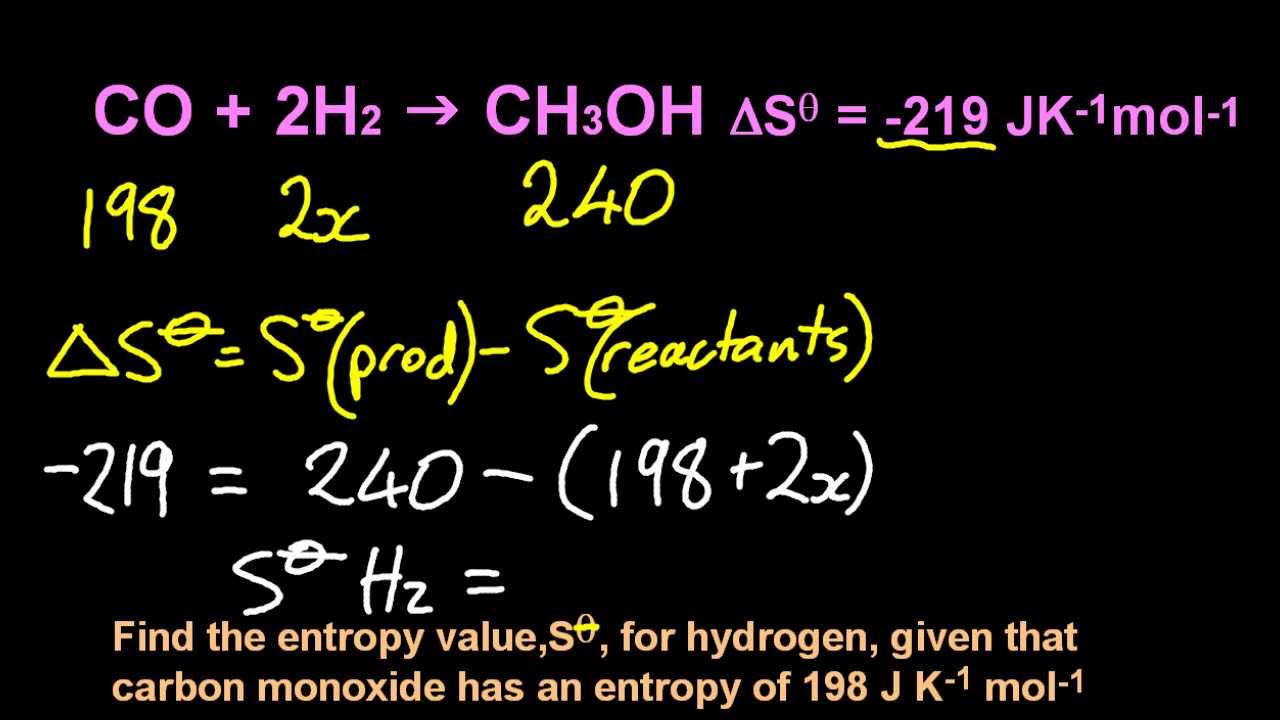
An analogous, although more complicated, equation can be written for a Newtonian (viscous) fluid in terms of viscosity and velocity gradients. However, that one regards change in entropy, not absolute entropy: dS dQ T (1) (1) d S d Q T. I’ve only seen two equations really relating to entropy thus far, and only one of them includes the temperature factor. This is the differential from of the heat conduction equation we all learned in freshman physics. I tried deriving a formula relating entropy (not change in entropy, but entropy itself) to temperature. Unified mechanics theory states that when a system undergoes thermodynamic change from state A to. 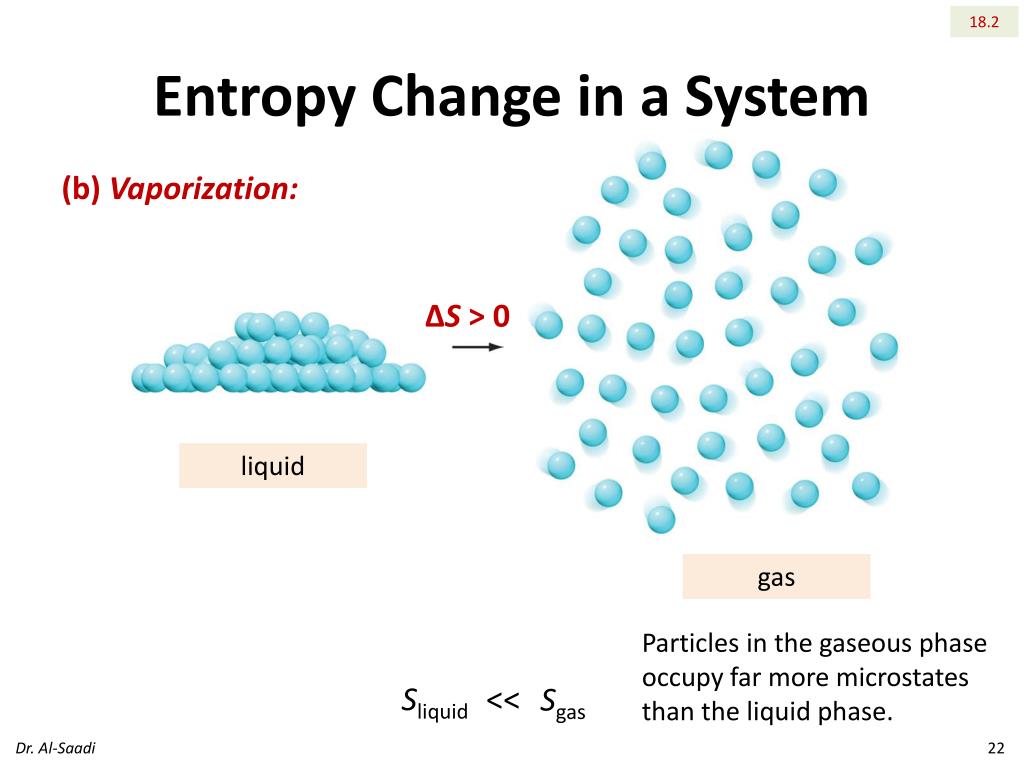
One definition of entropy change is $dS = \delta q_$$ where the value of k is measured or derived from molecular dynamics and statistical thermodynamics consideration. As is a measure of the total change in entropy at a point. The former is defined as a path where the system and its surroundings are in exact thermodynamic equilibrium at all points during the process. Two types of paths are defined: reversible and irreversible. The change in the universe is the sum of the changes in the system and its surroundings, so only two of the three are independent.Īny change in any thermodynamic state function is always independent of the path taken. You can calculate the entropy change of a reaction by subtracting the total entropy of the reactants from the total entropy of the products. The something of interest here is a thermodynamic state function of a system, its surroundings, or the universe. It means an infinitesimal change in something as it undergoes a process. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
